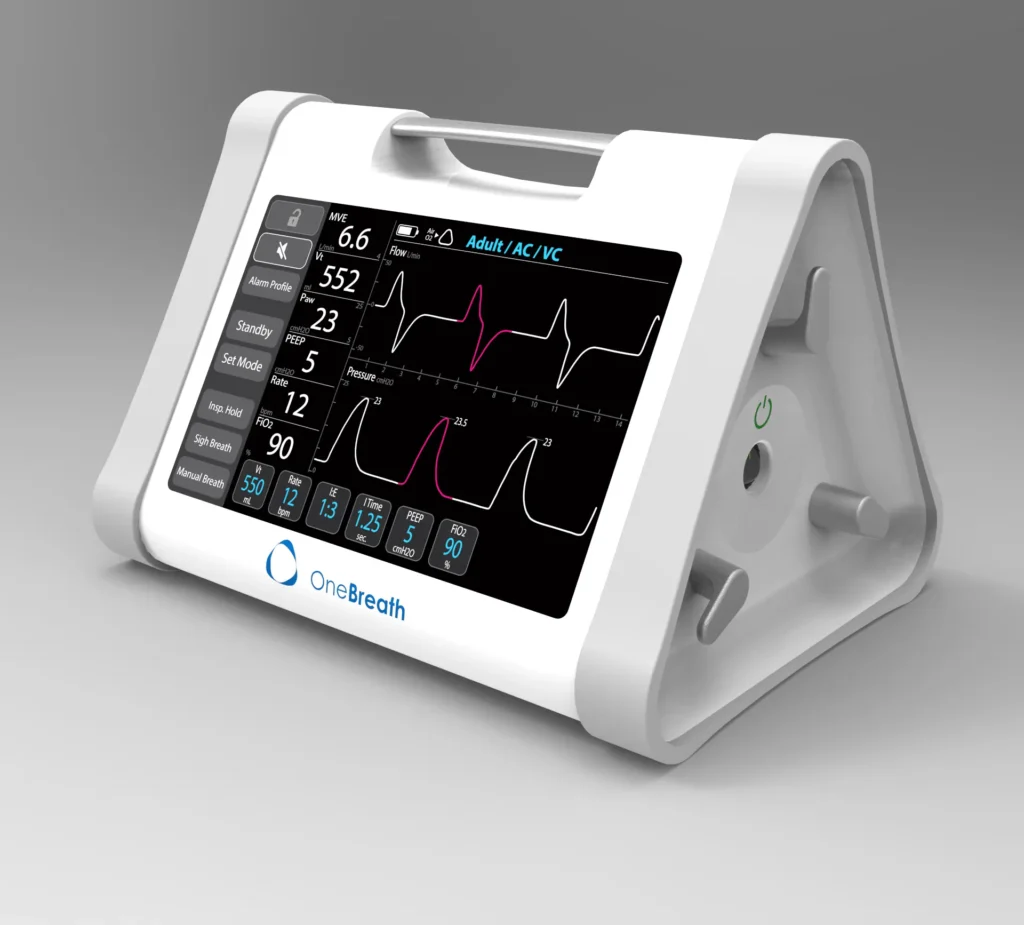
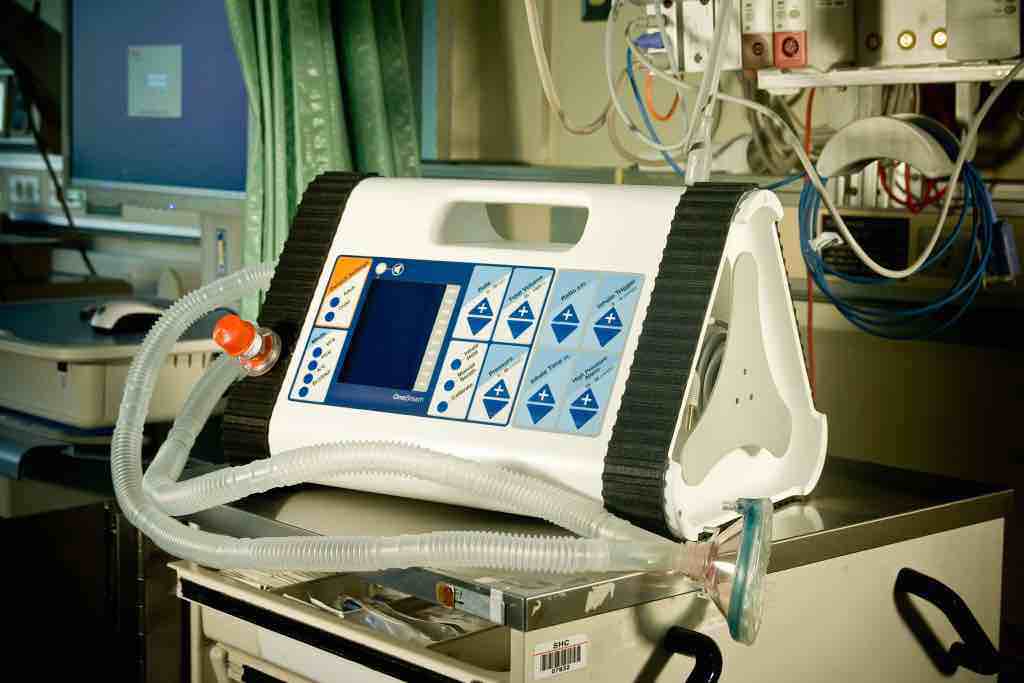
219 Design and Matt Callaghan teamed up to take the initial prototype from minimally viable to fully functional by performing extensive preliminary testing and global studies that ultimately improved the usability and aesthetics, increased functionality, decreased costs, and improved portability. The end result is a best-in-class ventilator sure to be a success across the globe.
Finding the Best Partner
Today, OneBreath is an international medical device company with a mission to advance healthcare in emerging countries by enabling their customers to incorporate world-class technologies using products designed for their unique environments. However, it all began with a vision to save lives by lowering costs.
When the world faced a global flu pandemic, Dr. Matthew Callaghan recognized that U.S. hospitals would experience a shortage of ventilators, resulting in lives lost. So, as a postdoctoral fellow in Stanford University’s biodesign program, he set out to address one of the most difficult problems in critical care medicine by designing a reliable, no-frills ventilator.
Dr. Callaghan wanted a single engineering firm that could handle all the project requirements. He also needed a company who believed in the concept enough to take on the project while funding was still limited. Finally, he wanted a true partner who shared his vision and passion for improving healthcare through technology and had the expertise across firmware, electronic engineering, mechanical and industrial design.
“Stanford Biodesign is a training incubator in medical technology that brings together multidisciplinary teams of medical, engineering, law and business school students to address unmet medical needs with innovative approaches.” – Stanford Medicine
Portable Ventilator for OneBreath
Designing For Global Use Through Immersion
Client Vision
To Advance Healthcare Through Accessible Technology

“219 Design was a great fit for OneBreath. Like biodesign, 219 applies an interdisciplinary approach to consistently produce thoughtful, unique, and innovative outcomes.”Matthew Callaghan, Founder and CMO
OneBreath
An Interdisciplinary Approach for Better Outcome
The proof of concept Dr. Callaghan brought to 219 Design required improved usability and aesthetics, increased functionality, component selection, and extensive preliminary testing. 219 Design tackled the project by putting together an interdisciplinary team that included product development, electronic and mechanical engineering, and medical expertise.
The collaboration resulted in two initial prototypes: works-like and looks-like. The team used these prototypes for preliminary user and human factors research at Stanford Hospital. There, top respiratory therapists provided feedback that resulted in a new prototype that met their desired specifications and requirements. It also resulted in an infusion of funding as the concept came closer to reality.
Popular Science Magazine
“The device is just a fraction of the cost of a low-end conventional ventilator, runs on a 12-volt battery for 6 to 12 hours at a time, and is smaller than a toolbox so it can be easily deployed wherever needed.”Popular Science Magazine
June 2010